|
The global genetic testing market is estimated to be valued at US$ 16.22 Bn in 2023 and is expected to exhibit a CAGR of 12.4% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights.
Market Overview: Genetic testing involves the analysis of an individual's DNA to identify genetic variations or mutations that may be associated with specific diseases or conditions. This market offers a wide range of tests including diagnostic tests, carrier tests, predictive tests, and presymptomatic tests. These tests provide crucial information for healthcare professionals and individuals, enabling personalized treatment and prevention strategies. The increasing demand for personalized medicine, advancements in technology, and the growing incidence of genetic diseases are driving the growth of the genetic testing market. Market key trends: One key trend in the genetic testing market is the increasing adoption of direct-to-consumer (DTC) genetic testing. DTC genetic testing allows individuals to access and analyze their own genetic information without involving healthcare professionals. This trend is driven by the availability of affordable testing kits and the convenience of accessing genetic information from the comfort of one's own home. However, this trend also raises concerns regarding the accuracy and interpretation of test results, as well as privacy and ethical issues. In conclusion, the global genetic testing market is poised for high growth due to the increasing demand for personalized medicine. The key trend of DTC genetic testing is expected to further drive market growth, although challenges related to accuracy, interpretation, and privacy need to be addressed. PEST Analysis: Political: The political factors that may impact the genetic testing market include government regulations and policies regarding genetic testing, privacy and data protection laws, and healthcare policies. Governments may impose restrictions or regulations on genetic testing to ensure ethical and responsible use of genetic information. Economic: The economic factors influencing the genetic testing market include healthcare expenditure, reimbursement policies, and the affordability of genetic testing services. Rising healthcare expenditure and favorable reimbursement policies can drive the growth of the market, making genetic testing more accessible to the general population. Social: Social factors such as increasing awareness and acceptance of genetic testing, growing interest in personalized medicine, and the rise in genetic diseases can drive the demand for genetic testing. As people become more aware of the benefits of genetic testing, the market is likely to expand. Technological: Technological advancements in genetic testing techniques and equipment, such as next-generation sequencing and non-invasive prenatal testing, are driving the growth of the genetic testing market. These technologies offer greater accuracy, efficiency, and cost-effectiveness, making genetic testing more accessible and attractive. Key Takeaways: The global genetic testing market is expected to witness high growth, exhibiting a CAGR of 12.4% over the forecast period (2023-2030). This growth can be attributed to increasing awareness and acceptance of genetic testing, advancements in technology, and rising healthcare expenditure. In terms of market size, the genetic testing market is projected to reach US$ 16.22 billion by 2023. This significant market size indicates the growing demand for genetic testing services worldwide. Regionally, North America is expected to be the fastest-growing and dominating region in the genetic testing market. This can be attributed to the presence of a well-developed healthcare infrastructure, favorable reimbursement policies, and increasing prevalence of genetic disorders in the region. Key players operating in the genetic testing market include 23&Me Inc., Qiagen NV, Eurofins Scientific, PerkinElmer Inc., Illumina Inc., Danaher Corporation, Myriad Genetics Inc., Abbott Laboratories, F. Hoffmann-La Roche Ltd., Quest Diagnostics Incorporated, BioRad Laboratories Inc., and DiaSorin S.p.A. These key players contribute to the growth of the market through product innovation, strategic partnerships, and expansions in different regions. Read More: https://www.marketwebjournal.com/genetic-testing-market-is-estimated-to-witness-high-growth/
0 Comments
The global Glycated Albumin Assay Market is estimated to be valued at US$ 571.0 million in 2023 and is expected to exhibit a CAGR of 8.07% over the forecast period from 2023 to 2030, as highlighted in a new report published by Coherent Market Insights.
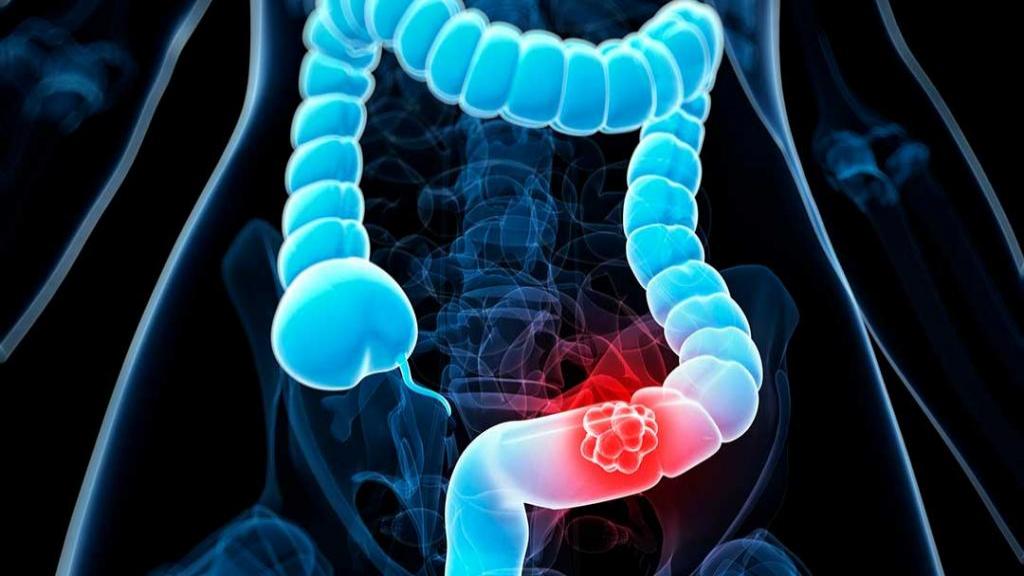
Market Overview: Glycated Albumin assay is a diagnostic test used to measure the levels of glycated albumin in the blood. Glycated albumin is formed when glucose molecules bind to serum albumin protein. The assay is primarily used for the diagnosis and management of diabetes. The advantages of the glycated albumin assay include its ability to provide a shorter-term indicator of glycemic control compared to other tests such as HbA1c, its sensitivity to changes in blood glucose levels, and its reliability in patients with certain conditions that can affect HbA1c levels. Market Key Trends: One key trend in the Glycated Albumin Assay Market is the increasing prevalence of diabetes worldwide. According to the International Diabetes Federation (IDF), in 2019, approximately 463 million adults (20-79 years) were living with diabetes, and this number is expected to rise to 700 million by 2045. This increasing prevalence of diabetes is driving the demand for glycated albumin assays as they provide a valuable tool in the diagnosis and management of the disease. Additionally, the glycated albumin assay offers advantages over other tests, such as HbA1c, making it an attractive option for healthcare professionals. In conclusion, the Glycated Albumin Assay Market is projected to witness high growth due to the increasing prevalence of diabetes and the advantages offered by the glycated albumin assay in the diagnosis and management of the disease. PEST Analysis: Political: The political environment can directly impact the Glycated Albumin Assay market. Government regulations and policies related to healthcare and diagnostic tests can affect the market's growth prospects. Changes in healthcare reimbursement policies and regulations regarding diagnostic tests may influence the adoption of Glycated Albumin Assays. Economic: The economic factors play a crucial role in shaping the market. Economic growth, healthcare expenditure, and per capita income affect the demand for diagnostic tests. The Glycated Albumin Assay market is expected to witness high growth due to increasing healthcare spending and rising prevalence of diabetes worldwide. Social: Social factors such as changing lifestyles, increasing awareness about preventive healthcare, and the aging population have a significant impact on the market. The growing prevalence of diabetes and the need for early diagnosis are driving the demand for Glycated Albumin Assays. Technological: Technological advancements in diagnostic tests and laboratory equipment have a strong impact on the market. The development of innovative and accurate assays enhances the efficiency and accuracy of diabetes diagnosis. The Glycated Albumin Assay market is witnessing advancements in technology, such as automated systems and point-of-care testing, which are driving market growth. Key Takeaways: The global Glycated Albumin Assay market is expected to witness high growth, exhibiting a CAGR of 8.07% over the forecast period. This growth can be attributed to the increasing prevalence of diabetes worldwide and the need for accurate and early diagnosis. The rising healthcare expenditure and growing awareness about preventive healthcare are also contributing to market growth. In terms of regional analysis, North America is expected to be the fastest-growing and dominating region. The region has a high prevalence of diabetes and a well-established healthcare infrastructure. The presence of key players and ongoing technological advancements in the healthcare sector are also driving market growth in North America. Key players operating in the Glycated Albumin Assay market include Asahi Kasei Pharma Corporation, Weldon Biotech Inc., Abbexa Ltd., Laboratory Corporation of America, Biocompare, EFK Diagnostics, Elabscience Biotechnology Inc., Abbott, Diazyme Laboratories Inc., Abnova Corporation, BSBE, Maccura Biotechnology Co., Ltd., LifeSpan BioSciences Inc., Biomatik, Geno Technology Inc., AMS Biotechnology (Europe) Limited, Epinex Diagnostics Inc., and DxGen Corp. These key players are actively involved in product development, collaborations, mergers, and acquisitions to strengthen their market position and expand their product portfolio. Read More: https://www.pressreleasebulletin.com/glycated-albumin-assay-market-size-and-share-analysis/ The global Colorectal Cancer Screening Market is estimated to be valued at US$ 12,866.16 Mn in 2021 and is expected to exhibit a CAGR of 7.5% over the forecast period 2023-2030, as highlighted in a new report published by Coherent Market Insights.
Market Overview: Colorectal cancer screening involves detecting precancerous polyps or colorectal cancer in the colon and rectum. The procedure helps in identifying abnormalities at an early stage, which improves the chances of successful treatment. Colorectal cancer is one of the leading causes of cancer-related deaths worldwide, and early detection through screening plays a vital role in reducing the mortality rate. Increasing awareness about the importance of early detection and prevention of colorectal cancer is driving the demand for screening procedures. Market Key Trends: One key trend in the Colorectal Cancer Screening Market is the adoption of non-invasive screening methods. Traditional screening methods, such as colonoscopy, have certain limitations and are associated with discomfort and invasiveness. Non-invasive screening tests, such as fecal occult blood tests (FOBT) and stool DNA tests, offer an alternative approach that is more convenient and less invasive for patients. These tests detect precancerous polyps or colorectal cancer by analyzing stool samples. The adoption of non-invasive screening methods is expected to increase due to their efficacy, ease of use, and patient preference. The Colorectal Cancer Screening Market is witnessing high growth due to increasing awareness about early detection and prevention of colorectal cancer. The adoption of non-invasive screening methods is a key trend in the market, driven by their convenience and patient preference. These trends are expected to fuel market growth over the forecast period. However, the market is highly competitive, with several key players operating in the industry, including Polymed PEST Analysis: Political: The political factors that may impact the colorectal cancer screening market include government regulations and policies related to healthcare, reimbursement policies for screening procedures, and initiatives by governments to increase cancer screening rates. For example, governments may introduce screening programs and provide funding for colorectal cancer screening, which can drive market growth. Economic: Economic factors such as healthcare expenditure, disposable income, and insurance coverage can influence the demand for colorectal cancer screening. Growing healthcare expenditure, rising disposable income, and increasing insurance coverage in many countries are expected to drive market growth. Additionally, the cost-effectiveness of screening tests may also impact market demand. Social: Social factors such as awareness about the benefits of cancer screening, changing demographics, and lifestyle habits can impact the market. Increasing awareness about the importance of early detection and screening for colorectal cancer, especially among high-risk individuals, can drive market growth. Moreover, changing lifestyles, including unhealthy diets and sedentary behaviors, may contribute to the increasing incidence of colorectal cancer, thereby increasing the demand for screening tests. Technological: Technological advancements in screening tests and diagnostic tools can significantly impact the market. Improved accuracy, sensitivity, and specificity of screening tests, as well as the development of non-invasive and convenient screening methods, can increase the adoption of colorectal cancer screening. For example, the introduction of multi-target stool DNA testing and blood-based tests for colorectal cancer screening has revolutionized the market. Key Takeaways: The global colorectal cancer screening market is expected to witness high growth, exhibiting a CAGR of 7.5% over the forecast period (2023-2030), due to increasing awareness about the benefits of screening and technological advancements in screening tests. The market size for 2021 was valued at US$12,866.16 Mn. Regionally, North America is anticipated to be the fastest-growing and dominating region in the colorectal cancer screening market. The region's strong healthcare infrastructure, high healthcare expenditure, and initiatives by governments to promote colorectal cancer screening contribute to its growth. Moreover, increasing awareness and favorable reimbursement policies are expected to drive market growth in North America. Key players operating in the colorectal cancer screening market include Polymedco Inc., Eiken Chemical Co. Ltd., Sysmex Corporation, Siemens Healthineers AG, Quidel Corporation, Novigenix SA, Hemosure Inc., Exact Sciences Corp., Epigenomics Inc., Olympus Corporation, and Clinical Genomics Technologies Pty Ltd. These companies are focused on developing innovative screening tests, expanding their product portfolios, and collaborating with healthcare providers to enhance their market presence. Overall, the colorectal cancer screening market is projected to experience substantial growth globally, driven by political initiatives, economic factors, social awareness, and technological advancements. North America is expected to lead the market, and key players are actively involved in product development and strategic collaborations to gain a competitive edge. Read More: https://www.newsstatix.com/colorectal-cancer-screening-market-is-estimated-to-witness-high-growth-owing-to-increasing-prevalence-of-colorectal-cancer-and-growing-awareness-about-early-detection/ The self-testing market has witnessed significant growth in recent years, driven by the rising demand for convenient and accessible healthcare solutions. However, this growth has brought forth its share of supply chain challenges that need careful navigation to ensure a seamless flow of products to consumers.
One of the primary supply chain challenges faced by the self-testing market is sourcing raw materials for manufacturing test kits. The production of self-testing kits involves the integration of various components, such as sensors, reagents, and packaging materials. Securing a steady supply of high-quality components while maintaining cost-effectiveness is crucial to the success of market players. Additionally, the transportation of self-testing kits from manufacturing facilities to distribution centers and retailers requires efficient logistics management. Delays or disruptions in the transportation process can lead to product stockouts or expiration of sensitive components, hampering market growth and customer trust. Regulatory compliance is another critical aspect of the self-testing market supply chain. With different countries and regions having specific rules and guidelines for medical device distribution, companies must navigate a complex web of regulations to ensure smooth product flow across borders. Moreover, the self-testing market faces challenges related to counterfeit products and unauthorized reselling. Ensuring the authenticity of self-testing kits throughout the supply chain is vital to protect consumers from inaccurate results and potential health risks. To address these challenges, market players are investing in supply chain optimization technologies. From implementing real-time tracking systems to using blockchain for enhanced traceability, these innovations aim to enhance transparency and accountability in the supply chain. The self-testing market must tackle several supply chain challenges to sustain its growth trajectory successfully. By focusing on efficient sourcing, robust logistics management, regulatory compliance, and anti-counterfeiting measures, industry stakeholders can overcome these hurdles and deliver reliable self-testing solutions to consumers worldwide. Consumer empowerment through self-testing has emerged as a transformative trend in the healthcare industry. With the proliferation of accessible and user-friendly self-testing kits, individuals now have the ability to take charge of their health proactively. Knowledge is the key aspect that underpins this movement, as informed consumers can make better decisions about their well-being.
Self-testing empowers consumers by providing them with valuable insights into their health status, ranging from basic indicators like blood pressure and glucose levels to more advanced genetic testing. By gaining a deeper understanding of their health, individuals can identify potential risk factors, track progress, and make lifestyle adjustments accordingly. This fosters a sense of responsibility for one's health, leading to a proactive approach in preventing illnesses and promoting overall well-being. According to Coherent Market Insights, the Global self-testing market is estimated to be valued at US$ 20,117.5 Million in 2022 and expected to reach US$ 39,175.0 million by 2030, witnessing a CAGR of 8.7 % over the forecast period (2022-2030). Furthermore, consumer empowerment through self-testing also extends to fostering patient-doctor collaborations. Armed with accurate test results, individuals can engage in more meaningful discussions with healthcare professionals, making consultations more efficient and focused. This collaborative approach enables better diagnosis, personalized treatment plans, and improved health outcomes. However, consumer empowerment through self-testing also comes with responsibilities. Ensuring test accuracy, interpreting results correctly, and seeking professional guidance when needed are essential aspects of this movement. Regulators and manufacturers must work hand in hand to maintain stringent quality control and education to safeguard consumers' health interests. In conclusion, consumer empowerment through self-testing is a promising trend that promotes a proactive approach to health management. Armed with knowledge, individuals can take better care of themselves, engage in informed conversations with healthcare providers, and make informed decisions about their well-being. Antimicrobial susceptibility testing (AST) plays a critical role in guiding appropriate antibiotic therapy. However, there are certain limitations associated with this testing method that need to be addressed. This blog post will explore the various challenges faced in antimicrobial susceptibility testing and discuss strategies and solutions to overcome them.
One major limitation is the time-consuming nature of traditional Antimicrobial Susceptibility Testing methods, which can delay the initiation of targeted therapy. Rapid diagnostic technologies, such as automated systems and molecular techniques, have emerged as potential solutions to expedite the testing process. This article will delve into the advantages and limitations of these novel approaches. Additionally, the emergence of multidrug-resistant pathogens presents another hurdle in Antimicrobial Susceptibility Testing. The blog will shed light on the strategies employed to overcome this challenge, including the development of new antimicrobial agents, combination therapies, and the use of alternative treatment options. By highlighting these limitations and proposing effective solutions, this blog post aims to foster discussions on improving antimicrobial susceptibility testing and ultimately contribute to better patient outcomes. Read More: https://cmitoc.blogspot.com/2023/06/antimicrobial-susceptibility-testing-is.html Government initiatives and policies play a crucial role in shaping the Colorectal Cancer Screening Market by promoting and facilitating screening programs, improving access to screening services, and implementing quality standards. Governments worldwide are recognizing the importance of early detection and prevention of colorectal cancer and are taking proactive measures to reduce the burden of this disease.
Many governments have introduced national screening programs or guidelines that recommend regular global colorectal cancer screening market for eligible individuals. These programs often provide subsidized or free screening tests, ensuring that cost is not a barrier to accessing screening services. Additionally, governments collaborate with healthcare organizations and advocacy groups to raise awareness and educate the public about the benefits of early detection. Moreover, regulatory policies are in place to ensure the quality and accuracy of colorectal cancer screening tests. Government bodies work closely with regulatory agencies to evaluate and approve screening technologies, ensuring their safety and efficacy before they are made available to the public. Government initiatives and policies have a significant impact on the Colorectal Cancer Screening Market by creating a supportive environment, driving demand, and fostering innovation in screening technologies. By prioritizing early detection, governments contribute to reducing the mortality and improving the overall health outcomes of individuals at risk of colorectal cancer. Read More: https://cmitoc.blogspot.com/2023/06/rising-prevalence-of-colon-cancer-to.html |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2023
Categories
All
|




 RSS Feed
RSS Feed
